Epigenetics of KSHV infection
One of our major goals is to investigate how specific viral and host factors can regulate the chromatin of the KSHV genome in infected cells. We are interested to determine how the modulation of the viral chromatin controls the primary infection, the establishment of viral latency, the maintenance of viral latency, and the lytic reactivation.
Chromatin states of the KSHV genome
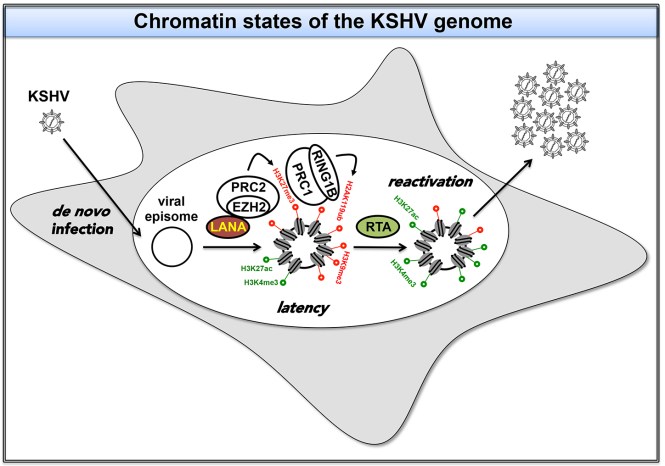
While the KSHV DNA is chromatin-free in the virion, it rapidly acquires a chromatin associated with specific histone modifications during de novo infection. The chromatinization of the KSHV genome is controlled by chromatin modifying enzyme complexes that are recruited onto the viral DNA in a spatially and temporally ordered manner. Following infection KSHV establishes latency in most cell types, which is characterized by the genome-wide inhibition of the expression of viral lytic genes.
Of the host epigenetic factors that are involved in the inhibition of lytic genes, the Polycomb Repressive Complexes (PRC1 and PRC2) are especially important, which are recruited onto the viral genome by the latent KSHV factor LANA in a stepwise manner. LANA can directly recruit the PRC2 complex whose enzymatic subunit EZH2 deposits the repressive histone mark H3K27me3 on the viral chromatin. Afterwards PRC1 is recruited to the viral chromatin through binding to H3K27me3 and the histone H2A E3 ubiquitin ligase RING1B of PRC1 deposits the repressive histone mark H2AK119ub on the viral chromatin. The polycomb proteins binding to the KSHV genome are crucial for the inhibition of lytic genes in infected cells and thereby for maintaining the viral latency.
Lytic reactivation from latency is mediated by the expression of the replication and transcription activator of KSHV (RTA), which results in the dissociation of PRCs from the promoter of lytic genes and inducing their expression. How RTA can reactivate KSHV from latency is still not fully understood.
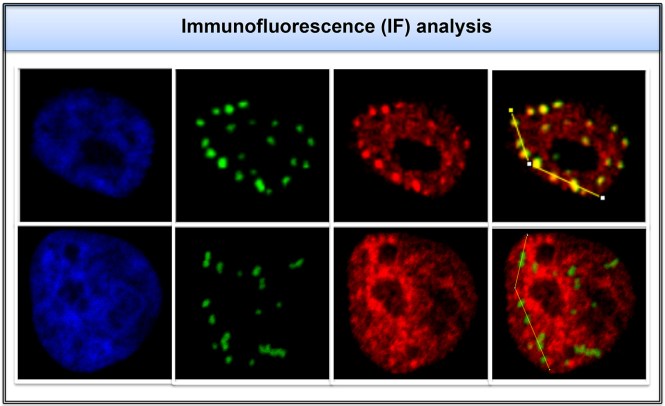
The KSHV epigenome in B cell lymphoma during viral latency and lytic reactivation
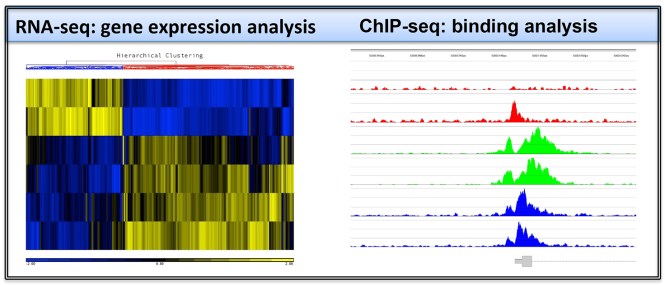
Primary effusion lymphoma cells (PEL) are B cell lymphoma cells that are persistently infected by KSHV. While PELs carry KSHV in latency, the lytic reactivation of KSHV can be triggered by RTA expression. We are using chromatin immunoprecipitation (ChIP) assays coupled with either DNA tiling microarray or next generation sequencing to study the role specific epigenetic modifications in viral gene regulation on the KSHV genome in PEL cells during both latency and lytic reactivation.

In collaboration with Dr. Bernadett Papp’s group, we have also started to interrogate the genome-wide transcriptional mechanisms and underlying epigenetic events leading to induction of host genes in PEL cells during viral reactivation. Our common collaborative goal with these genome-wide efforts is to identify novel regulators of the viral life cycle. Ultimately, by targeting our newly identified critical players, we hope to block viral reactivation and viral spreading.
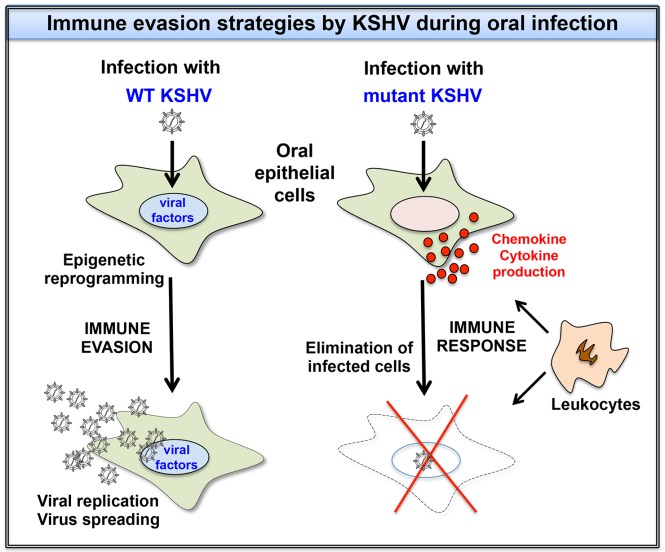
Immune evasion strategies of KSHV during oral infection
While the default pathway of KSHV infection in most cell types is the establishment of latency, KSHV has been shown to express lytic genes and replicate in oral epithelial cells following de novo infection. We are using KSHV mutants to determine what viral and host factors allow the lytic KSHV infection of oral epithelial cells. In this regard, we hypothesize that KSHV has to inhibit several immune response pathways during oral infection to be able to replicate, and we are interested to identify the underlying mechanisms. To begin to reveal these genome-wide early events during infections, Dr. Bernadett Papp’s group and my laboratory formed a highly active collaborative team to uncover the genome-wide host-pathogen interactions which can be critical during the initial step of infections.
Methods
Our collaborative research programs integrates reverse viral genetics with structural-functional biochemical assays, which are combined with system biology approaches to interrogate the function of viral and host factors involved in the epigenetic and transcriptional regulation of the KSHV genome and host genes that are critical for the persistent KSHV infection, KSHV-induced oncogenesis, and KSHV immune evasion.